Chromatography
Last Updated: Aug 6, 2013
Chromatography (Instrumental techniques in chemical analysis)
Chromatography is a broad range of method that is used to separate and analyze complex mixtures.. Chromatography is widely defined as the ability to separate molecules to make them remain in a stationary phase instead of a mobile phase. The molecules are separated using the partitioning characteristics of molecules.
Basic Terms Used In The Chromatography Technique
Chromatograph: -The instrument used in Chromatography.
Stationary phase: -Particularly solid or a highly viscous liquid. Stays in one place
Mobile phase: – Either liquid or gas. Moves along the column.
WORKING PRINCIPLE
The components in a mixture are separated based on their affinity to the mobile phase. The components having a different polarity and the mobile phase of a distinct polarity is passed through the column. Due to this one component will migrate faster than the other. Since the molecules of same compounds move in groups the compounds are separated to distinct bands within the column.
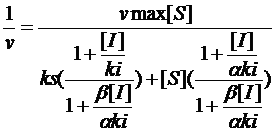
Step 1: The mixture of components remains in the top of the wet column
Step 2: While the mobile phase passes through the column, the two components starts to separate.
Step 3: In this figure the blue component has a stronger affinity (the tendency of a chemical substance to combine with another) for the mobile phase while the red component stays fixed in the stationary phase.
Step 4: each component is eluted (fluid exiting from the column) which is afterwards collected separately and analyzed.
Types of Chromatography:
- Partition chromatography: In this method both the stationary phase and the mobile phase are in the liquid form. The stationary phase is an immiscible liquid with the mobile phase.
2. Liquid-solid Chromatography: This method is similar to the partition chromatography except the fact that the stationary phase is replaced with a bounded silica or sometimes alumina inside the column. The components in the mobile phase having a greater affinity to the stationary phase will be absorbed into it and those that do not will pass through the column
3. Ion Chromatography: This method of chromatography is used to determine ions that are having a low ion exchange capacity. The equilibrium of the ions in the solutions and the counter ions to pair with the oppositely charged ions are fixed to the stationary phase.
Uses of Chromatography
- Can be used by pharmacists to analyze and determine the composition of chemicals found in each product.
- Can be used by the water supply authorities to determine the pollutants level in the drinking water
- Is used in TOXICOLOGY- that deals with identifying and separating drugs of abuse used for illegal purpose.
QUESTIONS AND ANSWERS
1. What is the difference between a stationary phase and a mobile phase?
Ans> The stationary phase are generally solids like silica gel or alumina whereas the mobile phase uses solvents. The solvent used as a mobile phase is absorbed by the stationary phase.
2. Which instrument is used in Chromatography?
Ans>The instrument used in Chromatography to carry out Chromatographic separations is called as Chromatograph. The Chromatograph consist of different entities like mobile phase supply system, the detector and the data processing system.
3. Which Chromatographic technique is most widely used?
Ans>The liquid chromatographic method is the most common and widely used method since it employs less expensive equipments.
Tell us Your Queries, Suggestions and Feedback
5 Responses to Chromatography
« Instrumentation and Application of IR WATER AND ITS INDUSTRIAL APPLICATION »


A very sophisticated topic to be written on. Article contains detail information about Chromatography and author did a great a great job by adding the essence of questions and answers. This will really help to know a lot about Chromatography.
the detail about chromatography is given here go through this
nice detailed,explantion of d topic,along with uses…q&a isnice to clear doubt…thank u
Clearly explained topic with illustrated figures. Easy to understand and informative. gud work 🙂
working of chromatography is explained in well mannered.and its application is also mentioned.