Combustion And Its Related Numericals
Last Updated: May 26, 2013
COMBUSTION is widely described as the process of burning of fuel and oxidant to produce heat. Oxidants are the substances that cause to combine with Oxygen. The process of combustion includes thermal, hydrodynamic and chemical process. The process starts like this:
- Fuel and oxidants are mixed in the presence of catalysts which are used to increase the rate of reaction.
- The fuel used can be either gaseous, liquid or solid
- The mixture is then induced with a heat source.
- The chemical reactions between fuel and oxidants take place.
- The heat released from this reaction creates a self-sustained process
- The resulting output of the combustion process results in heat, light, mechanical work and plasma.
Numericals on Combustion:
1. The combustion of methane in atmospheric air:-
the above equation is obtained after equating the molar coefficients from the reactants and products side.
The air-fuel ratio: it is defined as follows.
now comparing the reactants of the methane with the theoretical air we obtain:
2. The combustion of n-butane:
It is always found that combustion always occurs at elevated temperatures and all the products of combustion behave in ideal gas including water vapor.
During the analysis of the products of combustion the following things are the main areas of interest:
-> the percentage volume of specific products, particularly carbon dioxide and carbon monoxide.
->the dew point of water vapor; which requires the evaluation of partial pressure of water vapoure
The main purpose of combustion is to produce heat. The heat is produced through the change in enthalpy from the reactants to the products. The opposite of a combustion process is an adiabatic process where no heat is released. Due to this there is a significant increase in the temperature of combustion products.
The combustion of generic hydrocarbon fuels contains the oxidizer as oxygen which is found in atmospheric air. The combustion of solid fuels like kerosene occur in entirely different way.
Tell us Your Queries, Suggestions and Feedback
3 Responses to Combustion And Its Related Numericals
« Questions on Properties and Use of Terylene Who will police this police? »


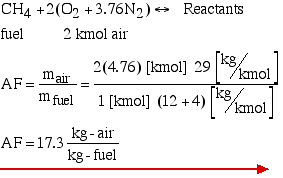

Nothing
Nothing
Combustion or burning is the sequence of exothermic chemical reactions between a fuel and an oxidant accompanied by the production of heat and conversion of chemical species.also numerical are given here.